Specific heat
Specific heat
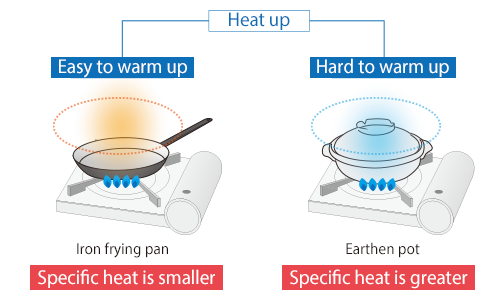
Specific heat is the amount of heat required for raising temperature of an object per unit mass by unit temperature. Its SI unit is [J/ (kg∙K)]. It is also called specific heat capacity.
An object with small specific heat can be heated or cooled quickly as its temperature changes by small amount of heat. On the contrary, heating or cooling an object with large specific heat takes longer.